prenatal screening program

Prenatal Care Provider FAQ
Frequently Asked Questions on the PNS Program
Program changes went into effect on September 19, 2022
NOTE: Kaiser providers should check with their internal systems to find out how ordering screening tests or result communications differs from what is summarized here.
Summary of Prenatal Screening Program Changes of 2022 (PDF, 2 pages)
Go to questions on:
Post PNS Program launch
Yes. As a result of a court ruling, a state regulation (17 CCR 6523(e)) that required clinical laboratories to have a contract with CDPH in order to offer or provide prenatal birth defect screening for fetal autosomal trisomies or neural tube defects will no longer be enforced. As a result, prenatal care providers may order these prenatal screening tests outside of the California Prenatal Screening (PNS) Program from any licensed clinical laboratory.
Prenatal care providers are still required to offer participation in the PNS Program to their pregnant patients. The PNS Program continues to provide California residents with access to high-quality screening for Down syndrome (trisomy 21), trisomy 18, trisomy 13, Turner syndrome, Klinefelter syndrome, trisomy X and XYY and MSAFP screening for neural tube defects for one low program fee as to each that is reimbursed by Medi Cal and most health insurance providers in the state. Only patients who elect to participate in the PNS Program will have access to free follow-up genetic counseling and diagnostic services at one of the many state-contracted Prenatal Diagnosis Centers located throughout the state for any pregnancy determined to be high-risk.
PNS Program participants with public or private insurance will not be subject to out-of-pocket copays and/or deductibles. Prenatal care providers are still required to offer participation in the PNS Program. Patients may choose not to participate in either type of testing offered through the PNS Program. If they choose not to obtain screening through the PNS Program, they may access screening outside of the PNS Program through any licensed clinical laboratory.
Prenatal care providers who order prenatal screening outside of the PNS Program will need to arrange for follow-up counseling and diagnostic testing for their patients outside of the state program if follow-up is necessary. Patients and their health insurance plans will likely be subject to higher prenatal screening and follow-up diagnostic fees compared with the PNS Program.
The PNS Program has not and does not prevent or limit providers from ordering screening tests not provided through PNS Program. Some of the current state-contracted screening laboratories allow the same blood draw used for the PNS Program to be used for such additional screening, but providers must complete and submit a separate test requisition form.
In keeping with recent published recommendations from the American College of Medical Genetics and Genomics (ACMG),
on April 1, 2024, the CA PNS Program added sex chromosome aneuploidies (SCAs), Turner syndrome, Klinefelter syndrome, Trisomy X and XYY.
Yes, California providers can order prenatal screening from any licensed clinical lab. As a result of a court ruling, a state regulation (17 CCR 6523(e)) that required clinical laboratories to have a contract with CDPH in order to offer or provide prenatal birth defect screening for fetal autosomal trisomies or neural tube defects will no longer be enforced.
However, for patients who elect to participate in the PNS Program, providers must order prenatal screening tests through the PNS Program from state-contracted labs.
Only patients who participate in the California Prenatal Screening (PNS) Program will have access to free follow-up genetic counseling and diagnostic services at a state-contracted Prenatal Diagnosis Centers for a pregnancy determined to be high-risk.
The PNS Program offers low program fees that are reimbursed by Medi-Cal and most California health insurance plans, with no cost sharing (copayment, coinsurance, or deductible).
Yes. The recording of part of the January 25, 2023 CalGenetic Portal training webinar is available.
Visit the PNS Program Portal Resources web page for additional videos on how to use the CalGenetic Portal.
1. Who can manage delegates (office staff with access to the Portal)
Four new types of professional roles are now able to add delegates in the CalGenetic Portal. The professions are licensed midwives, nurse practitioners, certified nurse midwives, and physician assistants. For licensed providers in these categories, they can now assign their own delegates in their own accounts. They first need to stop ordering tests as a delegate of another licensed provider, though. A user cannot be a licensed user and a delegate at the same time.
2. Unlinking a license from an account
Some providers have not been able to link their own medical license to their own account because staff in their office are already using that license number. A new self-service CalGenetic Portal feature allows a provider to unlink their license number from a previous account and create a new account linked to that license, without losing access to screening test orders and results, facility information, and delegate information.
Please see this recording from the PNS Program January 25, 2023 webinar addressing managing delegates and the new unlinking feature. See the Unlink License and Linking Orders Step-by-step Guide (PDF) for more details on how to use this feature.
Note that there is currently a defect that is affecting the “Unlink License” functionality in Firefox so please use Google Chrome or Microsoft Edge if you are experiencing issues.
1. Delegate access will be extended to 180 days from 90 days.
This change means that providers will need to re-approve delegate access only every six months, not three months. Note that the delegate’s access expiration date will remain the same, but the next time their provider renews their access, it will be renewed for 180 days (unless the provider changes it to a specific date on the Access Expires On field on the Delegate Profile).
2. A new patient result date field will be added to each screening order record.
In the CalGenetic Portal, the new date field on the screening order will show the date the patient result became available. This will help providers track resulted versus un-resulted mailers. This field will be sortable, so providers can see latest results at the top of the dashboard. A hack shared in the webinar is that providers can also search the dashboard for patient names using command+f on a PC and ctrf+f on a Mac.
Visit the PNS Program Portal Resources web page or CalGenetic YouTube playlist to view videos that provide step-by-step instructions for using the CalGenetic Portal to create a profile and view results for the PNS Program.
The videos explain how users can do the following:
- Providers create, access, and manage their user profile
- Providers create and manage their delegates’ access
- All users search, view, and download prenatal screening results
- Delegates create and access their user profile once an invitation from their managing provider is received
- All users order prenatal screening tests
The PNS Program label stationery can be ordered online. Please select the item called ‘PNS Program label stationery: Consent and Electronic Order Confirmation’ when you order your supplies.
Order the label stationery in its own order and you will receive a packet of at least 50 (maximum 300) within a day or two. There may be delays if you order other printed materials along with the special stationery if those other materials are not in stock.
If you are a member of a large clinical group, include your name within the ‘Organization/Department’ field on the online form. Direct the shipment to a specific staff-member by writing their name in the ‘Attention’ field. Only the ‘Organization’ name and ‘Attention’ person are shown on the shipping label with the address you provide.
Yes. Providers should place cfDNA or MSAFP screening orders using the CalGenetic Portal and print the screening Consent & Order Form using the PNS Program label stationery. This stationery contains the four peel-off stickers on the bottom of the form.
This stationery is available now and can be ordered through the PNS Program Supplies form. Please select the item called ‘PNS Program label stationery: Consent and Electronic Order Confirmation’ when you order your supplies.
When screening results are positive, a regional case coordinator will contact the provider, and the provider will receive the results by mail and in the CalGenetic Portal. The provider must alert the patient about the positive screening results and explain the availability of the PNS Program state-approved follow up services, including genetic counseling, ultrasound, and diagnostic testing.
No. As of February 1, 2023, if providers send the old First and Second Trimester test requisition form to the NAPS labs, the associated specimen will no longer be analyzed. Providers will receive a result mailer telling them to redraw their patient's prenatal screening specimen.
Providers should destroy all their old First and Second Trimester test requisition forms so they do not get used.
Providers should place cfDNA or MSAFP screening orders using the CalGenetic Portal and print the screening Consent & Order Form using the PNS Program label stationery. This stationery contains the four peel-off stickers on the bottom of the form.
This stationery is available now and can be ordered through the
PNS Program Supplies form. Please select the item called ‘PNS Program label stationery: Consent and Electronic Order Confirmation’ when you order your supplies.
Providers need to re-approve delegate access only every six months (180 days).
Visit the “contact us” section of the CalGenetic Portal or look for the contact information at the bottom of one of the patient screening result mailers.
To update your phone number for the multi-factor authentication (MFA):
1. Send an email to gdspadmins@cdph.ca.gov.
2. Subject line should be, “Update phone number for MFA.”
The email body must include full name, email address, current number on file, and new number you would like it changed to.
Yes. The patient’s insurance details must be entered when providers or delegates are ordering a screening test in the CalGenetic Portal. The information is required so that the PNS Program can bill the patient’s insurance for screening and should only be left blank if the patient does not have insurance.
Providers should do the following: (1) if relevant, contact the patient and request that they destroy the original order confirmation form, (2) initiate a new order in the CalGenetic Portal, (3) give the new order confirmation form to the patient to bring to the blood draw location. If the patient already has had the blood drawn using the first (inaccurate) order form, when the provider receives the test result, they should call their regional case coordinator (indicated on the bottom of the result mailer) to report the change so that an updated result can be re-issued.
Revvity (formerly PerkinElmer) began offering cfDNA lab services to all providers, not just Kaiser providers, on March 20, 2023.
Google Chrome, MS Edge, Firefox work best. Safari sometimes causes problems.
Some providers have not been able to link their own medical license to their own account because staff in their office are already using that license number. A new self-service CalGenetic Portal feature allows a provider to unlink their license number from a previous account and create a new account linked to that license, without losing access to screening test orders and results, facility information, and delegate information.
Please see this
recording from the PNS Program January 25, 2023 webinar addressing managing delegates and the new unlinking feature. See the
Unlink License and Linking Orders Step-by-step Guide (PDF) for more details on how to use this feature.
Note that there was a defect affecting the “Unlink License” functionality in Firefox. Please use Google Chrome or Microsoft Edge if you are experiencing issues.
PNS Program
Below is a checklist providers can use for getting grounded in the PNS Program.
| Steps | What to do | Where to do it |
|---|---|---|
| 1 |
Read Prenatal Care Provider FAQ web page to understand program details.
Watch the recorded webinar for more details on cfDNA |
PNS Program Provider FAQ web page (this page); |
| 2 | Watch CalGenetic Portal Training videos | PNS Program Portal Resources web page |
| 3 |
Register on CalGenetic Portal.
Ensure delegates are registered on CalGenetic Portal |
CalGenetic Portal |
| 4 |
Read state-contracted cfDNA lab instructions to understand blood collection details.
Order supplies directly from cfDNA laboratory, if needed |
PNS Program Information for Providers web page – See Program Materials section for cfDNA Lab Instructions Instructions for 2 labs are posted now |
| 5 |
Order Supplies. MSAFP blood draw kits, PNS Program label stationery and MSAFP mailing labels.
Order printed PNS Program educational materials (Provider Handbook, Patient Booklet, screen-positive booklets, etc.) |
PNS Program Supplies web page Be sure to use link for revised PNS Program, not the current program A nationide paper shortage is delaying the printing of some materials. Please download and print patient materials for program launch as needed (see Step 7 below). |
| 6 |
Download PNS Program Provider Handbook and
view its appendices online.
Bookmark PNS Program Information for Providers web page, to access resources such as patient back-up consent and decline forms |
PNS Program Information for Providers web page |
| 7 |
Download PNS Program patient educational materials (Patient booklet, etc.) from PNS Program website and
locate QR codes for the patient video in English and Spanish to share with patients |
PNS Program Education Resources - For Individuals and Families |
On September 19, 2022, the California Prenatal Screening (PNS) Program launched its revised program. Cell-free DNA (cfDNA) is now the primary screening test for trisomy 21, trisomy 18, and trisomy 13, replacing biochemical screening for trisomies 21 and 18.
cfDNA screening is available starting at 10 weeks 0 days and can be done throughout pregnancy. However, the PNS Program recommends that cfDNA screening take place from 10 weeks 0 days through 21 weeks 0 days so that all follow-up services can be available to individuals determined to be high risk. cfDNA screening also gives patients the option to find out the sex of the fetus. Providers can choose this option by checking a box when ordering screening. There is no additional cost for finding out the fetal sex.
In addition to cfDNA, maternal serum alpha fetoprotein (MSAFP) screening for neural tube defects is still offered, now from 15 weeks 0 days through 21 weeks 0 days of gestational age.
The PNS Program continue to provide follow-up genetic counseling and diagnostic services for participants with screen-positive results for the three trisomies or neural tube defects at state-approved Prenatal Diagnosis Centers.
Within the PNS Program, providers choose to work with one of the three state-contracted labs. They are Natera, Quest Diagnostics, and Revvity. They screen for trisomy 21, trisomy 18, and trisomy 13 and neural tube defects (what is on the state’s screening panel). The revised PNS Program screening panel does not currently include sex chromosome anomalies or microdeletions. See questions and answers below for more details on this.
Another change is the expanded use of the CalGenetic Portal for ordering screening and getting results. See questions and answers below for more details on this.
An additional change is regarding nuchal transluchency ultrasound. Nuchal translucency ultrasounds are still recommended as the most accurate dating method of gestational age. However, the PNS Program no longer requires a nuchal translucency measurement to receive a preliminary risk assessment for fetal chromosomal abnormalities with a first trimester biochemical screening test. The previous policy to authorize follow-up services at a state-contracted Prenatal Diagnosis Center with a large NT > 3.0 mm, regardless of the biochemical screening result, has ended.
However, if the cfDNA or MSAFP screening result is positive, and the patient goes to a state-contracted Prenatal Diagnosis Center and the fetus is identified to have a large NT > 3.0 mm, and the patient chooses to have an amniocentesis, the patient can choose to have a karyotype or microarray.
cfDNA screening through the revised PNS Program provides these benefits compared to biochemical screening:
- cfDNA provides higher sensitivity and lower false-positive rates, resulting in fewer screen-positive cases referred unnecessarily for follow-up diagnostic services;
- cfDNA can provide results for one additional aneuploidy (trisomy 13);
- the gestational age window will allow screening as early as 10 weeks of gestation; and
- prenatal providers will have a choice of more than one state-contracted cfDNA laboratory to conduct screening for patients.
Moving to cfDNA screening makes high-quality prenatal screening accessible for all pregnant individuals in California for the three most common autosomal trisomies at an affordable price.
In the PNS Program, the Genetic Disease Screening Program has contracted with the following three cfDNA laboratories only:
- Natera
- Quest Diagnostics
- Revvity
Natera and Quest Diagnostics completed the required onboarding process and are available for providers to choose in the PNS Orders Application on the CalGenetic Portal. Revvity became an available choice for providers on March 20, 2023. See below or visit the
PNS Program Information for Providers web page for specific cfDNA laboratory instructions.
Providers may send their patient samples to any of the three state-contracted cfDNA laboratories for the same program participation fee of $232.00.
- The cfDNA screen from Natera is performed using 10ml of whole blood collected in two cell-free DNA Streck tubes (20 ml total is required). These tubes are provided in Natera’s blood collection kit.
- The cfDNA screen from Quest Diagnostics is performed using 10 mL (8mL minimum) whole blood collected in one cell-free DNA Streck glass tube, if drawing blood outside one of the 400+ Quest Patient Service Centers.
Screening result communications and follow-up services will be standardized regardless of which state-contracted laboratory is selected.
Kaiser providers should check with their internal systems to find out how ordering screening tests or result communications differs from what is summarized here.
cfDNA Laboratory Instructions and FAQs
Natera
Quest
-
cfDNA Laboratory Instructions: Quest Diagnostics (PDF)
-
cfDNA Laboratory Additional Information: Quest Diagnostics (PDF)
Revvity
Yes, patient can opt to have fetal sex reported. Clinicians should talk to their patient to find out if they want fetal sex disclosed. Depending on the patient’s decision, clinicians should select the appropriate checkbox when ordering a cfDNA screening test. There is no additional cost for fetal sex reporting.
Starting April 1, 2024, the CA PNS Program will add sex chromosome aneuploidies (SCAs), Turner syndrome, Klinefelter syndrome, Trisomy X and XYY. The revised PNS Program screening panel does not currently include microdeletions. Providers can separately order these and other screenings, including carrier screenings, outside of the PNS Program screening panel.
Yes. Certain of the three state-contracted laboratories will allow providers to order additional screening for microdeletions using the same blood specimen taken from a patient through the revised PNS Program.
Even though the same blood specimen may be used, the additional screening at the state-contracted laboratory will technically be independent of the revised PNS Program. It will require the creation of a separate test request process. The laboratory conducting those tests will bill separately for these services.
Nuchal translucency ultrasounds are
still recommended as the most accurate dating method of gestational age. However, starting September 19, 2022, the PNS Program will no longer require a nuchal translucency measurement to receive a preliminary risk assessment for fetal chromosomal abnormalities with a first trimester biochemical screening test.
The previous policy to authorize follow-up services at a state-contracted Prenatal Diagnosis Center with a large NT > 3.0 mm, regardless of the biochemical screening result, has ended.
However, if the cfDNA or MSAFP screening result is positive, and the patient goes to a state-contracted Prenatal Diagnosis Center and the fetus is identified to have a large NT > 3.0 mm, and the patient chooses to have an amniocentesis, the patient can choose to have a karyotype or microarray.
Yes. The revised PNS Program only offers cfDNA screening for singleton and twin gestations. Patients with multiples of three or more, or twin gestations with a demise, are not candidates for the PNS Program. Similarly, patients with fetal demise are not candidates for the PNS Program.
Patients that are not candidates according to the PNS Program’s cfDNA screening criteria should not participate in the PNS Program. For these patients who are not candidates for the PNS Program, their prenatal care provider may seek alternative options for the care of these patients, such as referral to a Maternal Fetal Medicine specialist.
Cell-free DNA (cfDNA) screening is now established as the most reliable and accurate prenatal screening method for the three most common autosomal aneuploidies: trisomy 21 (Down syndrome), trisomy 18, trisomy 13, and the four most common sex chromosome aneuploidies, Turner syndrome, Klinefelter syndrome, trisomy X and XYY. Due to the improved screening performance of cfDNA, which includes lower false positive rates, the state decided to stop using biochemistry to screen for fetal chromosome abnormalities. Maternal serum alpha fetoprotein (MSAFP) screening will continue to be offered to screen for neural tube defects (NTDs) in the second trimester.
Yes. PNS case coordinators support prenatal screening providers with program implementation and case management.
PNS case coordinator offices are located throughout California. Each prenatal screening provider is assigned a PNS case coordinator by zip code. The prenatal screening provider should rely on the PNS case coordinator as the primary source of information regarding the PNS Program. The PNS case coordinator’s phone number is included on every result mailer.
A PNS case coordinator will contact a provider office when there is missing or incomplete information on a screening test order or to verify information for certain results such as positives, samples drawn too early or too late, or any non-negative screening test result.
CalGenetic Portal
Yes. The CalGenetic Portal is live. It is accessed at https://calgenetic.cdph.ca.gov/. Providers should only use this url address and check that any previously set up bookmarks point to this address only.
Authenticated users can already obtain secure online access to do the following:
1) View patient prenatal screening results. The PNS Program will continue to mail results but accessing them on the CalGenetic Portal is quicker and easier for providers.
2) Submit Outcome of Pregnancy forms. These data are used to evaluate screening performance, and all data requests should be now entered on the CalGenetic Portal.
3)
Order Screening Tests. A new electronic order requisition application on the CalGenetic Portal is now available. The CalGenetic Portal is used to order both the cfDNA screen and the MSAFP screen. Users must select the correct screen (cfDNA or MSAFP) when submitting an order. Each screen has a different ordering process. Providers will be required to obtain a signed patient consent document for each both screening tests, which must accompany the sample sent to the testing laboratory.
All physicians and their delegates are encouraged to log into the CalGenetic Portal and set up an account now. See the next question and answer in this FAQ or visit the PNS Program Portal Resources web page for a chart on who can access the Portal.
The chart below shows access to the CalGenetic portal, by profession. Note: Delegates are all staff members, including office and medical assistants, that a licensed clinician (MD/DO) allows access to the CalGenetic Portal.
|
Professions |
All access with license verified by IDEAL* |
Can access results as licensed users? |
Can place orders as licensed users? |
Can place orders as delegates? |
Can add delegates? |
Can Add Account Admin/Can Delete Account Admin |
Can be added as Account Admin |
|---|---|---|---|---|---|---|---|
| Physicians and Surgeons |
Yes |
Yes |
Yes |
- |
Yes |
Yes |
Yes |
| Osteopathic Physicians and Surgeons |
Yes |
Yes |
Yes |
- |
Yes |
Yes |
Yes |
| Licensed Midwife |
Yes |
Yes |
Yes |
- |
Yes |
Yes |
Yes |
| Physician Assistant |
Yes |
Yes |
Yes |
- |
Yes |
Yes |
Yes |
| Nurse Midwife |
Yes |
Yes |
Yes |
- |
Yes |
Yes |
Yes |
| Nurse Practitioner |
Yes |
Yes |
Yes |
- |
Yes |
Yes |
Yes |
| Registered Nurse |
Yes |
Yes |
- |
Yes |
No |
No |
Yes |
| Temporary Registered Nurse License |
Yes |
Yes |
- |
Yes |
No |
No |
Yes |
| Clinician Nurse Specialist |
Yes |
Yes |
- |
Yes |
No |
No |
Yes |
| Public Health Nurse |
Yes |
Yes |
- |
Yes |
No |
No |
Yes |
| Genetic Counselors |
- |
Yes |
- |
Yes |
No |
No |
Yes |
| Postgraduate Training License |
Yes |
Yes |
Yes |
No |
Yes |
Yes |
Yes |
| Office and Medical Assistants |
- |
- |
- |
Yes |
No |
No |
Yes |
* Interoperability Development Effort to Authenticate Licensees (IDEAL) is used by Medical Board of California to authenticate and support access to the CalGenetic Portal
Visit the PNS Program Portal Resources web page or CalGenetic YouTube playlist to view videos that provide step-by-step instructions for using the
CalGenetic Portal to create a profile and view results for the PNS Program. The videos explain how users can do the following:
- Providers create, access, and manage their user profile
- Providers create and manage their delegates’ access
- All users search, view, and download prenatal screening results
- Delegates create and access their user profile once an invitation from their managing provider is received
- All users order prenatal screening tests
Starting September 19, 2022, providers should order both the cfDNA and MSAFP screening tests using the CalGenetic Portal.
After a screening order is electronically submitted on the CalGenetic Portal, providers will need to print a PNS Program “Consent and Order Confirmation” for each cfDNA and MSAFP screen, respectively. The “Consent and Order Confirmation” will need to be printed on PNS Program label stationery provided at no cost.
The stationery can be ordered now on the
PNS Program supplies order web page. The PNS Program label stationery contains four peel-off unique barcode number labels that should be affixed to the required sample collection tubes and to the required paperwork for cfDNA screening and MSAFP screening orders.
Yes. Providers should order supplies on the PNS Program supplies order web page. The supplies available include the PNS Program label stationery, MSAFP mailing labels, and MSAFP blood collection kits.
PNS Program label stationery. Providers should order both the cfDNA and MSAFP screening tests using the CalGenetic Portal. After a screening order is electronically submitted on the CalGenetic Portal, providers will need to print a PNS Program “Consent and Order Confirmation” for each cfDNA and MSAFP screen, respectively.
The “Consent and Order Confirmation” will need to be printed on PNS Program label stationery provided at no cost. The PNS Program label stationery contains four peel-off unique barcode number labels that should be affixed to the required sample collection tubes and to the required paperwork for cfDNA screening and MSAFP screening orders.
MSAFP mailing labels. If you do not ship your specimens to our laboratories via General Logistics Systems (GLS), you will need to preprint the USPS pre-paid MSAFP mailing label and affix it to the MSAFP blood collection kit so that it is sent to the appropriate state-contracted Newborn and Prenatal Screening (NAPS) Laboratory.
In the past, the mailing label was included on the first trimester and second trimester TRFs.
The PNS Program is now providing these blank MSAFP mailing labels for printing at no cost. Order them through our
PNS Program supplies order web page as you would other supplies.
Providers that have already placed orders for MSAFP mailing labels and have yet to receive their supply should do the following in the meantime: Request an emergency packet from the PNS Program by emailing PNS@cdph.ca.gov and using the subject line, “Mailing Labels Request.”
MSAFP blood collection kits. These include yellow-top collection tubes, bio-hazard wrappers, and plastic trays. MSAFP screening requires blood to be drawn in a 3 mL serum separator tube provided in the state MSAFP blood collection kits. As before, after blood is collected from non-Kaiser patients for MSAFP screening, it should be sent to one of three NAPS (Newborn and Prenatal Screening) Laboratories.
Note that providers can also place orders now for revised PNS Program educational materials, but delivery of these materials may be delayed by a nationwide paper shortage.
Yes. The PNS Program has posted back-up, “Consent and Order Forms” as fillable pdf files for both cfDNA and MSAFP screening on the
PNS Program Information for Providers web page (see the Program Materials section). See also consent language translated into other langauges provided as a courtesy there.
Clinicians will be responsible for planning ahead by downloading and printing paper copies of the “Consent and Order Form” pdfs to use as backup forms if the CalGenetic Portal cannot be accessed. Note that patient “Decline” forms have been posted on the same web page.
If and when the pdf of the “cfDNA Consent and Order Form” or the “MSAFP Consent and Order Form” is downloaded and printed for use, a signed copy should be placed in the patient's health record and the signed original handed to the patient and included with their blood collection kit when blood is drawn either in your office or clinic, or at a specified blood collection facility. Note that clinicians need to tell patients where the closest blood collection facility is.
The patient’s blood sample and paperwork will all get shipped to the selected cfDNA laboratory. The selected laboratory performs the test and sends the screening results electronically to the PNS Program.
The signed patient “Decline” forms should be placed in the patient’s health record.
Not always. Kaiser providers should check with their internal systems to find out how ordering screening tests or result communications differs from what is summarized here.
Providers will continue to obtain results through the
CalGenetic Portal. The PNS Program will also continue to send result mailers by UPS ground mail to the patients’ prenatal care provider indicated on the PNS Program order form.
Providers should contact their patients with their screening results.
Yes. Clinicians and their delegates should place orders only for the patients for whom they are responsible for providing prenatal care.
Clinicians do not need to renew their registration for the
CalGenetic Portal. Their license is authenticated every time they log into
CalGenetic Portal.
Delegates are staff members, including office and medical assistants, that a licensed clinician (MD/DO) allows access to the CalGenetic Portal. Delegates generally do not have a professional license and therefore do not go through IDEAL verification on the CalGenetic Portal.
Staff, including office and medical assistants, can order screening tests on behalf of a physician or osteopath if they work for them. The staff member must be added as a delegate by the clinician on the CalGenetic Portal, and the clinician must indicate that the delegate can order screening tests in the CalGenetic Portal.
Will it be possible to order screening tests through EPIC rather than through the CalGenetic Portal?
No. Currently, there is no integration with EPIC given the diversity of institutions across the state and electronic platforms. Clinicians will need to use the
CalGenetic Portal to submit orders and obtain results for cfDNA and MSAFP prenatal screening. In the future the program is interested in partnering with large health care systems to better integrate PNS Program screening orders with electronic medical record systems.
Yes. The cfDNA or MSAFP screening test results will available through the Results Application on
CalGenetic Portal. However, prenatal providers must have the PNS Program form number of the original order and the patient’s date of birth to retrieve results on the Results Application in the
CalGenetic Portal. Results are available online for one year from the date of blood collection.
Using the Orders Application in the CalGenetic Portal has the following benefits:
- Licensed providers and delegated staff can keep track of specimens.
- Clinicians and delegated staff review updates on specimen status (e.g., “specimen received” and “specimen results”) on the orders inventory page.
- Clinicians and delegated staff navigate to results directly from the orders page.
- Clinicians and delegated staff will not have to wait to receive paper mailers to obtain results.
- Order submission takes less time with the copy and paste function: Order information that was submitted for one screening test (i.e., cfDNA) can be copied and used to submit the next screening test (i.e, MSAFP) for the same patient.
- Orders can be drafted before a patient’s arrival, and the order will remain in “draft” status until the order is ready to be submitted.
- Ordering providers can easily update their profile information, especially their address which is where result mailers are sent.
Yes. Delegates are staff members, including genetic counselors, office assistants, and medical assistants, that a licensed clinician (MD/DO) allows access to the CalGenetic Portal.
Staff, including genetic counselors, and office and medical assistants, can order screening tests on behalf of a physician or osteopath if they work for them. The staff member must be added as a delegate by the clinician on the CalGenetic Portal, and the clinician must indicate that the delegate can order screening tests in the CalGenetic Portal. The clinician can add the genetic counselor’s existing account’s email address as a delegate or add a new account belonging to the genetic counselor.
Additionally, genetic counselors can authenticate through the CalGenetic Portal based on their own license to view screening test results. They can also become delegates under a clinician’s license to order screening tests from the CalGenetic Portal using the same account, once added by the clinician.
The electronic Orders Application in the CalGenetic Portal shows the status for an order being processed. The status will be one of the following: “Draft,” “Submitted,” “Not received,” “Specimen Received,” “Specimen Processed,” and “Result Available.”
Orders can be drafted before a patient’s arrival, and the order will remain in “Draft” status until the order is ready to be submitted. Once submitted, the status is changed to “Submitted,” and then, “Not Received,” which means it has not yet arrived at the designated lab. After it is received at the lab, the status changes to “Specimen Received” and then subsequently, “Specimen Processed,” and lastly, “Result Available.”
Results should be available for viewing in the CalGenetic Portal 10–14 days from original blood draw date. If you cannot view results within that time frame, please contact your regional case coordinator office to get your patient’s result.
If a provider gets a message when registering as a user in the CalGenetic Portal that says “License Already in Use,” it means that the license number is already linked to an account (possibly staff in the same clinic are using it).
A new self-service CalGenetic Portal feature allows a provider to unlink their license number from a previous account and create a new account linked to that license, without losing access to screening test orders and results, facility information, and delegate information.
Please see this recording from the PNS Program January 25, 2023 webinar addressing managing delegates and the new unlinking feature. See the Unlink License and Linking Orders Step-by-step Guide (PDF) for more details on how to use this feature.
Note that there is currently a defect that is affecting the “Unlink License” functionality in Firefox so please use Google Chrome or Microsoft Edge if you are experiencing issues.
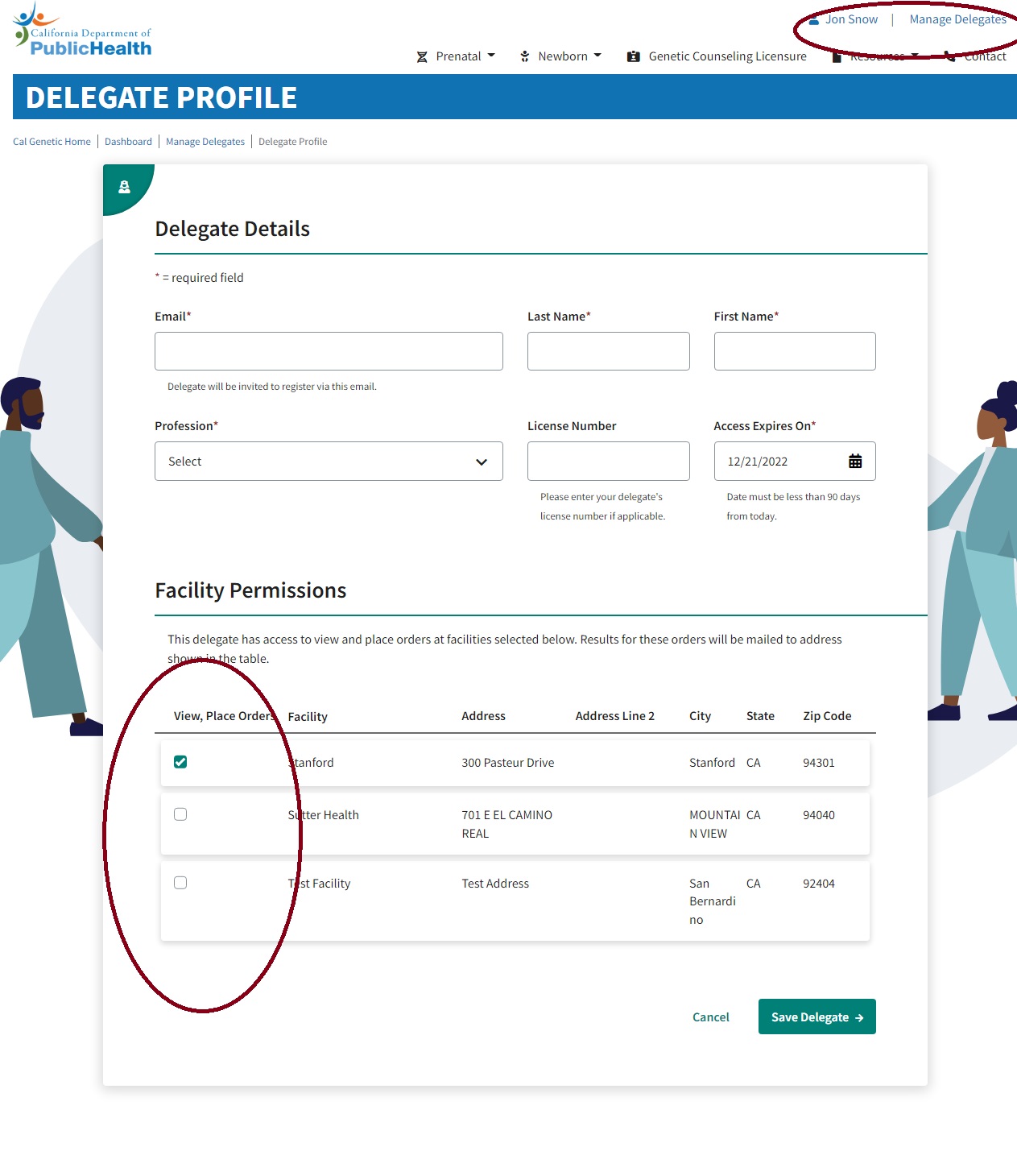
The screenshot below shows how the providers can give permission to a delegate (any office staff) to place orders and view results. This video also shows how to add or update delegates (see the timestamp at 3:26).
As the provider is adding or updating a delegate, they can select by checking the facilities for which they want them to place the orders. To reach the relevant screen in the CalGenetic Portal, please follow the below steps:
- Login to CalGenetic Portal
- Click on “PNS Results” or “PNS Orders” from the dashboard
- Click on the user name in the header and navigate to provider profile
- Add facility
- Add/update delegate
- If the delegate already exists, click on the delegate name in the grid. A single delegate can now be associated with multiple providers.
Changing fetal sex disclosure from “no” to “yes” after placing an order:
cfDNA screening
For the PNS Program, regarding cfDNA screening, providers will have a choice of three state-contracted cfDNA laboratories (Natera, Quest Diagnostics, and Revvity) to choose when ordering the cfDNA screening test through the CalGenetic Portal.
Regarding MSAFP screening, similar to how it is under the current program, specific state-contracted laboratories are assigned for the MSAFP screening based on the zip code location of the provider. Providers will be able to go to the
CalGenetic Portal to enter the zip code and preprint MSAFP mailing labels that can be affixed to the MSAFP blood collection kits for their patients. The MSAFP mailing labels and other supplies should be ordered on the
PNS Program supplies order web page.
All pregnant individuals in California can participate and receive cfDNA and MSAFP screening through the PNS Program regardless of age or risk. See the most recent ACOG bulletin covering prenatal screening, ACOG Practice Bulletin 226: Screening for Fetal Chromosomal Abnormalities. (Obstet Gynecol, Vol 136, No 4, October, 2020).
It states that “Prenatal genetic screening (serum screening with or without nuchal translucency [NT] ultrasound or cell free DNA screening) should be discussed and offered to all pregnant patients regardless of age or risk for chromosomal abnormality.”
The PNS Program makes cfDNA screening for trisomy 13, trisomy 18, and trisomy 21 available to all pregnant individuals regardless of age or risk.
If the cfDNA laboratory cannot provide a definitive result, the result mailer will explain the specific patient result. In some cases, a redraw will be recommended. Screening test failures due to low fetal fraction will be eligible for redraw if the patient’s gestational age is less than 18 weeks; if the gestational age is 18 weeks or greater, the patient will be eligible for a redraw or referral for counseling, ultrasound and diagnostic testing at a state-contracted Prenatal Diagnosis Center.
Will the PNS Program share information about how to work with the three state-contracted cfDNA labs?
Yes. See below for the laboratory-specific instructions for each respective state-contracted cfDNA. These instructions are also posted on the PNS Program Information for Providers web page (see the Program Materials section).
cfDNA Laboratory Instructions and FAQs
Natera
Quest
-
cfDNA Laboratory Instructions: Quest Diagnostics (PDF)
-
cfDNA Laboratory Additional Information: Quest Diagnostics (PDF)
Revvity
-
cfDNA Laboratory Instructions: Revvity (PDF)
-
cfDNA Laboratory Additional Information: Revvity (PDF)
These documents include laboratory-specific instructions, where to send patients for cfDNA blood collection, and how individual provider offices can obtain cfDNA blood collection kits, prepare samples, and ship samples.
These directions are also posted on the
PNS Program Portal Resources web page and will be on the CalGenetic Portal resource page at program launch.
Yes. cfDNA screening requires blood to be drawn in a 10 mL Streck tube – otherwise known as a “tiger top,” or black and white top tube.
- The cfDNA screen from Natera is performed using 10ml of whole blood collected in
two cell-free DNA Streck tubes (20 ml total is required). These tubes are provided in Natera’s blood collection kit.
- The cfDNA screen from Quest Diagnostics is performed using 10 mL (8mL minimum) whole blood collected in
one cell-free DNA Streck glass tube, if drawing blood outside one of the 400+ Quest Patient Service Centers.
Yes. The prenatal screening test with a screen positive result must be done through the PNS Program, and the patient must meet screening criteria for follow-up services, in order for the patient to be eligible for PNS Program follow-up services.
cfDNA screening will be available starting at 10 weeks 0 days and can be done throughout pregnancy. However, the PNS Program recommends that cfDNA screening take place from 10 weeks 0 days through 21 weeks 0 days so that all follow-up services can be available to individuals determined to be high risk.
What are the advantages or disadvantages to choosing one state-contracted laboratory over the other?
Each of the state-contracted labs provide comparable screening performance with respect to screening for the three most common fetal autosomal trisomies (trisomy 21, trisomy 18 and trisomy 13). However, the process for obtaining screening for patients is different for each lab.
For Natera, providers will need to order blood collection kits and can draw blood in their own office or send the patient to a Natera contracted draw station.
For Quest Diagnostics, patients can be sent directly to one of their 400+ Patient Service Centers.
Please visit our
PNS Program Information for Providers web page to see the laboratory-specific instructions for ordering cfDNA screening. Please also review the “additional Information” document for each laboratory, which provides more specific details for each laboratory.
Please see this list of
Reference List on cfDNA Screening (PDF) that was created as part of the April 18, 2022, webinar on the Clinical Overview of cfDNA Screening. Please visit the
PNS Program Webinars web page to view the recording of that and other program change webinars.
Both cfDNA and MSAFP screening
Nuchal translucency ultrasounds are still recommended as the most accurate dating method of gestational age.
The PNS Program will no longer require a nuchal translucency measurement to receive a preliminary risk assessment for fetal chromosomal abnormalities with a first trimester biochemical screening test, however. The previous policy to authorize follow-up services at a state-contracted Prenatal Diagnosis Center with a large NT > 3.0 mm, regardless of the biochemical screening result, has ended.
However, if the cfDNA or MSAFP screening result is positive, and the patient goes to a state-contracted Prenatal Diagnosis Center and the fetus is identified to have a large NT > 3.0 mm, and the patient chooses to have an amniocentesis, the patient can choose to have a karyotype or microarray.
Prenatal screening for twins will be provided through the revised PNS Program. Pregnancies with more than two fetuses are not eligible for prenatal screening through the revised PNS Program. SCA detection via cfDNA screening in the California PNS Program is only available for monozygotic twins. SCA screening is not available for multiple gestation of two (dizygotic twins) or more fetuses.
Fetal reduction or fetal demise will make a patient ineligible for prenatal screening through the revised PNS Program. A list of all conditions that would make a patient ineligible for screening is provided on the
CalGenetic Portal and is displayed when a provider begins the screening orders process.
For MSAFP screening, there were only a few changes in September 2022. The state continues to supply complete MSAFP blood collection kits, which include yellow-top collection tubes, bio-hazard wrappers, and plastic trays.
What is different? If you do not ship your specimens to our laboratories via General Logistics Systems (GLS), you will need to preprint the USPS pre-paid MSAFP mailing label and affix it to the MSAFP blood collection kit so that it is sent to the appropriate state-contracted Newborn & Prenatal Screening (NAPS) Laboratory.
In the past, the mailing label was included on the first trimester and second trimester test request forms (TRFs). The PNS Program provides these blank MSAFP mailing labels for printing at no cost. Please note that there is no special printer required to print the new labels. You can use any ink jet or laser jet.
You can order the mailing labels using the same mechanism you currently use to order PNS Program supplies.
Yes. The patient can opt for one screening test but not the other through the state program. Both screening tests are recommended because they screen for different birth defects however.
Providers can order the one screening test through the CalGenetic Portal and print the online decline form to decument that the patient has declined the other.
If a patient declines one or both screening tests made available through the PNS Program, the patient will not be able to get a screening test outside of the PNS Program for what is on the PNS Program screening panel (trisomy 21, trisomy 18, trisomy 13, and neural tube defects).
Decline forms are available on the PNS Program Information for Providers web page (see the Program Materials section) for either test for providers to use to keep track of the patient’s decision not to get one or both of the screening tests. The decline form should be signed by the patient and put in the medical record.
Yes. General Logistics Systems (GLS) is still available for shipping specimens to NAPs laboratories. Providers can set up an account to have specimens picked-up and delivered through Golden State Courier (GLS) free of charge by calling (916) 542-9390 or emailing
govtaccts@gls-us.com.
What is different? If providers do not ship specimens to our laboratories via GLS, they will need to preprint the USPS pre-paid MSAFP mailing label and affix it to the MSAFP blood collection kit so that it is sent to the appropriate state-contracted Newborn & Prenatal Screening (NAPS) Laboratory. In the past, the mailing label was included on the first trimester and second trimester TRFs. The PNS Program provides these blank MSAFP mailing labels for printing at no cost. Please note that there is no special printer required to print the new labels. You can use any ink jet or laser jet.
Providers can order the mailing labels using the same mechanism currently used to order PNS Program supplies.
Yes. In our current program, maternal serum alpha fetoprotein (MSAFP) screening for neural tube defects has been offered from 15 weeks 0 days through 20 weeks 0 days of gestational age. For MSAFP screening in the revised PNS Program, screening for neural tube defects will continue to be offered, now from 15 weeks 0 days through 21 weeks 0 days of gestational age.
Patient billing and payment
As of September 19, 2022, the PNS Program participation fees is
$232 for cfDNA screening and
$85 for MSAFP screening. The cfDNA screening fee will increase to
$344 on July 1, 2024. The PNS Program participation fee is for comprehensive screening and follow-up, meaning that if a patient has a positive screening result, the fee will cover all follow-up services including genetic counseling, ultrasound, and diagnostic testing.
Per state regulations,
Medi-Cal and private insurance must cover all program fees, with only a few exceptions for self-insured employers and out-of-state health plans. Patients are directly billed if they do not have insurance or if their insurance is affected by these exception categories and won't cover fees.
Per state regulations, Medi-Cal and private insurance must cover all program fees, with only a few exceptions for self-insured employers and out-of-state health plans. Patients are directly billed if they do not have insurance or if their insurance is affected by these exception categories and won't cover fees. Blood draw fees are not included in the program fees, however.
The PNS Program invoices the $344 PNS Program fee for the cfDNA screening or the $85 PNS Program fee for MSAFP screening to the patient’s insurance company. Providers do not need to do any billing. The PNS Program also provides direct payment to the state-contracted cfDNA laboratories.
PNS Program Participant Billing and Payment web page has more details about patient billing and payment. Please share this web page with patients wanting to know more about billing and payment.
Yes. If the patient is pregnant and pending Medi-Cal, the PNS Program fees will be covered retroactively.
Medi-Cal and private insurance will not automatically cover blood draw fees or any co-pays associated with blood draw for the revised PNS Program. The patient should check with their insurance. Blood draw fees are usually under $6.
The PNS Program screening panel does not currently include sex chromosome abnormalities or microdeletions. In some instances, pregnant individuals will need screening for sex chromosome abnormalities, whether or not they participate in the PNS Program. Screening for sex chromosome abnormalities is a service outside of the PNS Program and is not covered by the current PNS Program participation fee. It will require the creation of a separate test request process.
Billing for this service should use CPT code ‘81479,’ Unlisted molecular pathology procedure. Claims should be submitted to the patient’s insurance company with the medical indication for the procedure and a description of the procedure performed so that accurate pricing may be applied. The cfDNA laboratory conducting those tests will bill separately for these services.
No. Providers do not need to do any billing for the PNS Program. The PNS Program invoices the $232 PNS Program fee for the cfDNA screening or the $85 PNS program fee for MSAFP screening to the patient’s insurance company. The PNS Program also provides direct payment to the state-contracted cfDNA laboratories. Any previous arrangements between cfDNA laboratories and insurance companies or health care plans will no longer apply.
PNS Program educational materials
The California Prenatal Screening Program held webinars on cfDNA screening. The recorded webinars are posted on the
PNS Program Webinars web page. This web page allows for viewing 1.5 hour past webinar recordings, filling out CME evaluation surveys for live or recorded webinars, learning about how to get CME credits, and learning who can earn them. The first webinar on cfDNA screening is “cfDNA Screening: A Clinical Overview,” and features Dr. Mary Norton from UCSF.
Take note of or bookmark the PNS Program Information for Providers web page. It is the web page that has links to the
CalGenetic Portal and every program material or resource providers will need to access for the revised PNS Program launch, including the back-up order forms, the decline forms, the cfDNA lab instructions and links to the supplies ordering, webinar, and portal resources web pages.
To receive notices of PNS Program changes electronically before the new program launch, providers and other can
sign-up to receive the PNS Program e-Newsletter. Providers or delegates who register on the CalGenetic Portal will also receive the e-newsletter.
Education Resources for Individuals and Families web page houses patient materials for the revised PNS Program, along with the following:
Genetic Support Foundation Videos
PNS Booklet
Prenatal Screening Patient Booklet - English (PDF, 1.3MB)
- Armenian (PDF)
- Chinese, Simplified (PDF, 24.4MB)
-
Farsi (PDF)
-
Russian (PDF)
- Korean (PDF)
- Spanish (PDF)
- Tagalog (PDF)
-
Vietnamese (PDF)
- Hmong (PDF)
- Punjabi (PDF)
Prenatal Screening Patient Booklet X and Y Insert - English (PDF, 2 pages)
Patient Booklet Summaries - These summaries should be used in addition to the patient booklets, not instead of them. It is important that each patient receive the patient booklet. The patient booklet includes a full description of the program and its components, what consent means, and mandated legal notices.
- English (PDF)
-
Armenian (PDF)
-
Chinese, Simplified (PDF)
- Farsi (PDF)
-
Hmong (PDF)
-
Korean (PDF)
- Punjabi (PDF)
-
Russian (PDF)
-
Spanish (PDF)
-
Tagalog (PDF)
-
Vietnamese (PDF)
Prenatal Screening Patient Glossary - English (PDF, 2 pages)
Prenatal Screening Patient Booklet (use “select language” button in top right of web page to view in additional languages)
Screen Positive Booklets
-
Trisomy 21 (Down Syndrome) (PDF)
- Trisomy 18 (PDF)
- Trisomy 13 (PDF)
- Neural Tube Defects (PDF)
- Turner Syndrome (PDF)
- XYY (PDF)
- Trisomy X (PDF)
- Klinefelter Syndrome (PDF)
Other
Prenatal Screening Patient Video (English, MP4, 3.29 min)
Sexual Orientation and Gender Identity (SOGI) Information Survey
- QR code for Patient Video in English (save and share with patients to use with a QR Code Reader)

- QR code for Patient Video in Spanish (save and share with patients to use with a QR Code Reader)

- QR code for Patient Video in Chinese (save and share with patients to use with a QR Code Reader)

To download, distribute, or share a PNS Program patient video file, please follow a link to the vimeo site and download each video separately.
In addition to this FAQ: the PNS Program Information for Providers web page; the PNS Program e-newsletter, along with monthly program change update e-mail blasts; and the PNS Program Changes webinar series.
The following materials are also available on the Education Resources for Healthcare Providers webpage:




